|
Hydrogen is an important special case with a single valence electron and a valence of 1. For examples, carbon has a valence of 4 and has 4 valence electrons, nitrogen has a valence of 3 and has 5 valence electrons, and oxygen has a valence of 2 and has 6 valence electrons. carbon through neon), the valence of each atom plus the number of electrons in the valence shell in that atom always equals eight. Next we discover that there is a pattern to the valences: for elements in groups 4 through 8 (e.g. This systemization is quite remarkable and is very useful for remembering what molecules may be easily formed by a particular element. Hence, for many elements in the periodic table, the valence of its atoms can be predicted from the number of steps the element is away from the nearest inert gas in the table. The two groups of elements immediately after the inert gases, the alkali metals and the alkaline earths, have valences of one and two, respectively. NH3, PH3), and the elements before that (carbon and silicon most notably) have valences of four (CH4, SiH4 The elements just preceding these (nitrogen, phosphorus, antimony, arsenic) have valences of three (e.g. The elements just prior to the halogens (oxygen, sulfur, selenium, tellurium) are each two steps away from the inert gases in the table, and each of these elements has a valence of two (e.g. This "one step away, valence of one" pattern can be extended. Each inert gas is immediately preceded in the table by one of the halogens: fluorine precedes neon, chlorine precedes argon, bromine precedes krypton, and iodine precedes xenon. The inert gases with a valence of 0 sit to one side of the table. 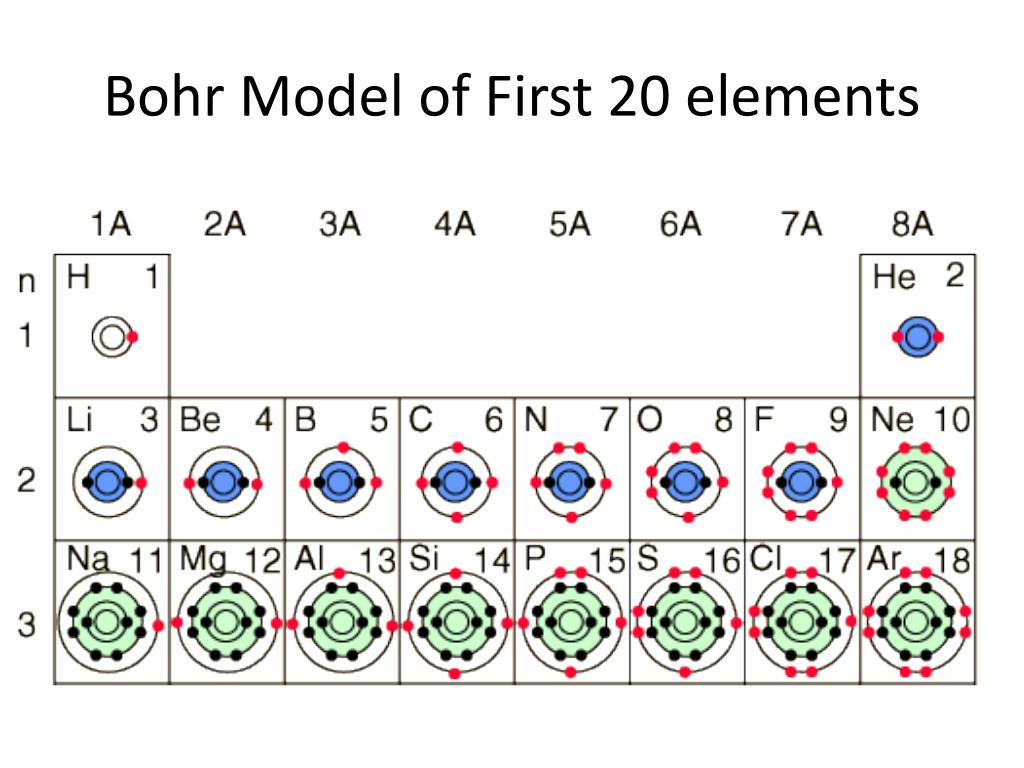
In doing so, we discover that the periodic table is a representation of the valences of the elements: elements in the same group all share a common valence. We can proceed in this manner to assign a valence to each element, by simply determining the number of atoms to which this element's atoms prefer to bind. Correspondingly, it is not surprising to find that, for example, the combination of two potassium atoms with a single oxygen atom forms a stable molecule, since oxygen's valence of 2 is be satisfied by the two alkali atoms, each with valence 1. Therefore, they also have a valence of 1. 
Lithium, sodium, potassium, and rubidium bind with a single halogen atom. This concept also applies to elements just following the inert gases. have a valence of 3, and carbon and silicon have a valence of 4. is assigned as 2, since two hydrogens are required to satisfy bonding needs of these atoms. What we mean by a valence of 1 is that these atoms prefer to bind to only one other atom. We thus assign their valence as 1, also taking hydrogen to also have a valence of 1. Each halogen prefers to form molecules by combining with a single hydrogen atom (e.g. We thus assign their valence as 0, meaning that these atoms tend to form 0 bonds. The inert gases do not tend to combine with any other atoms. To begin our analysis of chemical bonding, we define the valence of an atom by its tendencies to form molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
